EFFECT OF LACTOBACILLUS CASEI SHIROTA IN PREVENTING ANTIBIOTIC ASSOCIATED DIARRHOEA INCLUDING CLOSTRIDIUM DIFFICILE ASSOCIATED DIARRHOEA IN PATIENTS WITH SPINAL CORD INJURIES: A MULTICENTRE RANDOMISED, DOUBLE-BLINDED, PLACEBO-CONTROLLED TRIAL.
Naveen Kumar (Principle Investigator), Joy Chowdhury, Aheed Osman, Theresa Garrett (Research Nurse) and Jayne Edwards (Research Nurse)
Midland Centre for Spinal Injuries (MCSI), Oswestry
Funding / Sponsor: Yakult Honsha Co Ltd (YHL)
STUDY GROUP: National Spinal Injuries Centre, Stoke Mandeville Hospital; School of Health Science, City, University of London; Norwich Medical School, University of East; The Princess Royal Spinal Injuries Centre, Northern General Hospital and Yakult Honsha European Research Centre for Microbiology, Belgium.
Background: Antibiotic associated diarrhoea (AAD) and Clostridium difficile infection (CDI) are of major clinical concern in spinal cord injury (SCI) rehabilitation.
Methods: A multicentre, randomized, placebo-controlled, double-blind trial was conducted to assess (1) the efficacy of consuming a probiotic containing at least 6.5×109 live Lactobacillus casei Shirota (LcS) in preventing AAD and CDI; (2) whether undernutrition risk and proton pump inhibitors (PPI) are risk factors for AAD / CDI; (3) associated changes in quality of life and (4) the detection of C. difficile, its toxin type, and associated changes in other microbiota. LcS / placebo was given once daily for the duration of an antibiotic course and continued for 7 days thereafter. This trial was registered with ISRCTN: 13119162.
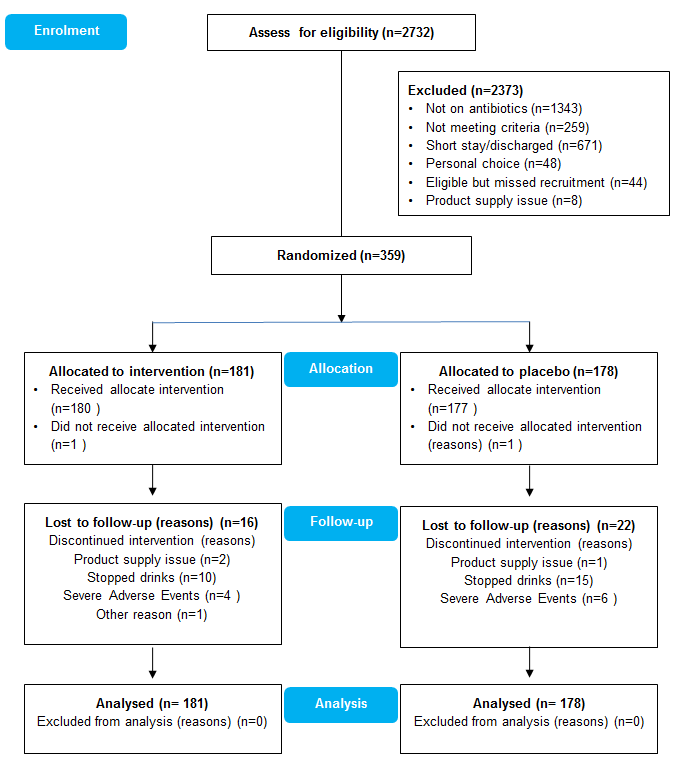
Findings: 359 consenting patients (median age: 53.3; range: 18-88 years) with SCI (49.4% tetraplegic) with a requirement for antibiotics due to infection were randomly allocated to receive LcS (n=181) or placebo (n=178). Overall, no statistical difference in occurrence of AAD or CDI between two groups (AAD, 7d: 32.5% v 31.6%, p=0.86; 30d: 45% v 42.1%, p=0.57; CDI: 7.1% v 3.7%, p=0.22). However in the participants regularly taking a PPI, LcS was found to prevent AAD at 7 days (19% v 35.7%, p=0.041) and at 30 days (28% v 47.8%, p=0.016). The relative risk for AAD in PPI-subgroup was 053 (0.29 to 0.99); 0.53 (0.32 to 0.91), and the number needed to treat was 6 (3.1 to 101); 4.2 (2.3 to 19.2) at 7-day and 30-days respectively. C. difficile was detected in 72 samples from 719 patients (A+B+ type: 57; A-B- type: 15) by qPCR. Focusing on the analysed combinations of stool samples, temporal gut microbiota alpha-diversity change, there were no significant differences in alpha-diversity metrics between two groups.
Interpretation: The present study indicates that LcS could not prevent AAD / CDI in unselected SCI patients but has the potential to prevent AAD in the higher risk group of patients on PPI. Confirmatory studies are now needed to allow translation of this apparent therapeutic success into improved clinical outcomes.
Enrolment summary:
UK multicentre, randomized, controlled study evaluating efficacy of LCS in prevention of antibiotic acquired diarrhoea (AAD) in spinal cord injury patients. MCSI joined this research (ISRCTN 13119162, MREC N°14/SC/1101,UKCRN ID 17618) in 2014.
For the ECLISP study 57 patients completed the study from Midland Centre for Spinal Injuries based at RJAH Orthopaedic and District Hospital NHS Foundation Trust, Oswestry.
Picture that shows MCSI Research team ( Mr Naveen Kuamar, Teresa Garret) in a final/closure meeting held in Stoke Mandeville with international ECLISP study investigators who successfully completed the ECLSIP esearch project: